Bulbitech featured in exclusive life science report: Unlocking potential in Norway
Dennis Hens, the Dutch CEO of Norwegian neuro-opthalmology device player Bulbitech, outlines the company’s journey to market, how its technology can support both optometrists and neurologists in their work, and gives an overview of the investment landscape for innovative medtech in Norway.
 Can you outline the technology behind Bulbitech and the problem you are trying to solve?
Can you outline the technology behind Bulbitech and the problem you are trying to solve?
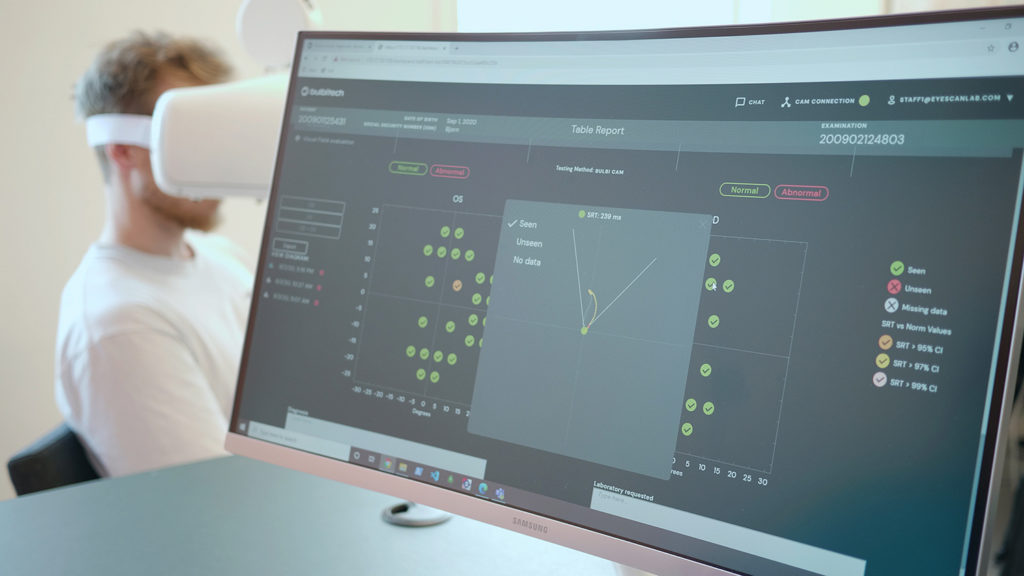
DENIS HENS (DH): We offer diagnostic support through eye tracking, especially in the neuro-ophthalmic landscape. When a patient goes to the hospital, often there is not a single neuro-ophthalmologist on duty or, having travelled more than four hours to get to the hospital, there are long waiting times.
Our eye-tracking camera – Bulbicam – connects to a working station on which our Bulbihub software can be used and attempts to solve this problem. This hardware-software combination can be deployed anywhere; the system is based in Northern Norway, but the outcomes and data can be shipped to a neuro-ophthalmologist in Trondheim or in Oslo. This creates an extended arm for neuro-oph- thalmologists, making neuro-ophthalmology available to patients in a local setting. Patients will no longer have to travel for hours just to take a diagnostic test; something that no longer makes sense today when everybody can be con- nected remotely.
We have developed three tests for which Bulbihub can be used: a visual field test based on eye movement perimetry; a pupil test, because we know a lot of glaucoma patients have pupil defects; and a ptosis test which is related to the fact that droopy eyelids can be a symptom of glaucoma. These three tests allow for early stage screening for glaucoma, something that both optometry and ophthalmology currently focus a lot on.
How easy is it to secure investment in Norway where medtech is relatively underdeveloped? What kind of education to investors require about Bulbitech and its technology?
DH: Norway is a very good country to start projects. In the pre-seed and seed stage people here are very investment-minded and there are a lot of angel investors and well organised angel networks who, once they understand a problem and can envision a solution, are willing to commit. There is also a good national incentive to invest, with tax returns on investments, which makes the pre-seed and seed stage a very attractive investors landscape.
However, at later stages of a company’s devel- opment things get more difficult. Norway only has a population of five million and there are not a lot of specialised medtech and biotech venture capitalists (VCs) or private equity. Therefore, most major investments are made through investment bankers with international networks or via syndication partners. There are a few good potential investors in this field but finding investment can feel like looking for a needle in a haystack.
Additionally, many of the investment banks that we talk to are willing to invest in biotech or medtech but have traditionally been focused on Norway’s more well-established industries of forestry, agriculture, fishing, and oil and gas. Therefore, there is a need to educate them about the steps that need to be taken to become a mature medtech company. That is something that could really be improved in Norway.
The Nordic countries have a strong research footprint neuro-ophthalmology. How is Bulbitech collaborating with consortiums or research groups in the region and using its geographic position to its advantage?
DH: It was difficult for us to enter the clinical landscape in Norway but fortunately now we are in talks with some good partners, including Oslo University Hospital, which is interested in our technology, and Helgeland Hospital in Northern Norway, with whom we are on the verge of signing a cooperation agreement.
We have also recently been approached by a neurologist working at St. Olav’s University Hospital, the neurological centre of which is world-renowned and has won a Nobel Prize. Their work is very different from what we gen- erally do – winning a Nobel Prize for research with mice is far from employing a diagnostic device in human beings and doing research with that – but once we have passed CE approval, these kinds of collaborations will become much easier.
Prior to the COVID-19 pandemic diagnostics were an often-overlooked part of the life sciences value chain. Do you sense this shifting and how are you planning to raise awareness of the importance of diagnostics in neuro-ophthalmology?
DH: There have certainly been a lot of therapeutics developed in recent years – rightfully so – but for patients to access therapeutics, they first need technology to define exactly what their needs are; something which can only be done through correct diagnosis. Diagnostics has been somewhat overlooked but that does not mean that it cannot be improved; I believe that with the right approach, diagnostics companies can be very interesting, especially in those areas where there is a shortage of certain medical professionals, such as neuro-ophthalmology. With the rising demand of an ageing population and a shortage of professionals, technology can step in and fill the gaps in both diagnostics and therapeutics.
Big Pharma will be a driving force behind this shift. Due to the substantial amounts of capital that exists there, they can gain wider acceptance of the importance of diagnostics than diagnostics companies alone can. In our case, Big Pharma is increasingly interested in eye movement perimetry, so there are multiple opportunities for collaboration and growth.
What is the potential for Bulbitech in terms of partnerships with Big Pharma companies focusing on ophthalmology treatments?
DH: There is a lot of potential. For instance, research shows that some early stage drugs against Alzheimer’s or Parkinson’s have a positive or negative effect on eye movement, which our device is more than capable of detecting. Diagnostics companies like ours have a huge opportunity to partner up with pharma companies developing such drugs for both the eye and the brain. Our focus tends to be more on the physiological side of the eye and the processes that occur when a person interprets data or sees lights, which affect eye movements.
Where do you see Bulbitech in five years’ time? Where do you want to lead it?
DH: We want to be in a position where eye doctors and opticians are familiar and comfortable with implementing eye tracking in their diagnostic processes. As part of our five-year plan we want to be selling in India and China, which will mean that we have conquered the markets of Europe, North America, and Asia. That is where we want to be, but before that we have the responsibility to educate people on how to use eye movements and our technology in optometry and ophthalmology diagnostics.
